Physics-Informed Endovasculature Deformation Estimation And Registration
Research at CERLAB (master's thesis)
project status: ongoing
I’m working on remote surgical robotics at the Computational Engineering & Robotics Lab at CMU under Dr. Kenji Shimada & doctoral candidate Rishi Basdeo at the Department of Mechanical Engineering, with collaborators and clinicians from University of Pittsburgh’s Surreality Lab and neurosurgeons from UPMC.
My focus is on the computer vision subsystem — building a full pipeline from physics-informed synthetic data generation through deep network training for intra-operative vessel deformation prediction.
We’ve also completed a comprehensive literature review in the realm of robotic-assisted endovascular surgery, which I’ve written the vision sub-system section of (currently under review at Springer Nature’s Journal of Intelligent and Robotics Systems).
the problem
During endovascular procedures, a guidewire is navigated through blood vessels under live X-ray (fluoroscopy). As the wire advances, it deforms the vessel wall. Predicting how the vessel deforms — before it tears — requires knowing the 3D displacement of the vascular centerline from a 2D X-ray projection. This is an inherently ill-posed inverse problem.
synthetic data pipeline
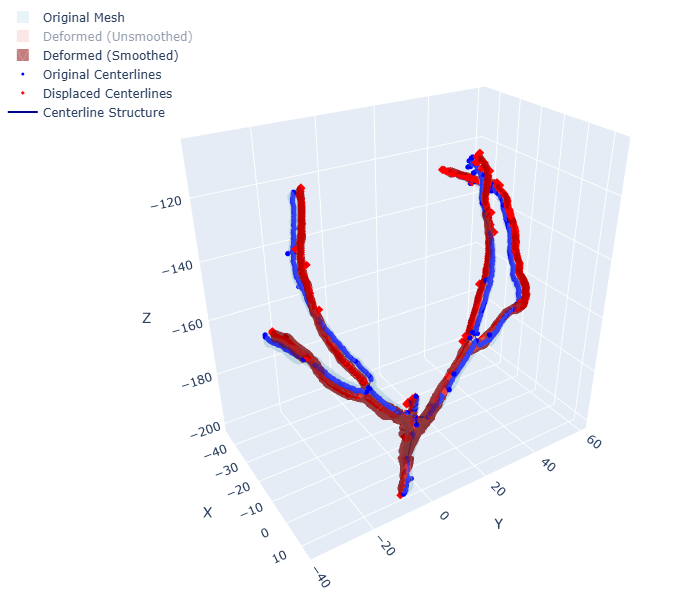
Since ground-truth intra-operative 3D vessel deformation data doesn’t exist at scale, I generate it synthetically using physics simulation.
Each simulated frame yields:
- Sparse 3D displacement fields — ground-truth deformation vectors at 20 control points along the centerline
- Multi-view synthetic X-rays — rendered via
gVirtualXRayacross 6 clinically relevant projections:AP,Lateral,LAO45,RAO45,Spider, andRAO30_Caudal20 - Physics metrics — per-frame max displacement (mm), mesh surface strain ratio, and vessel straightening percentage
I built an auditing tool to validate the physics integrity of the entire dataset, verifying that mesh strain stays within the clinical safety threshold of 1.15 (a proxy for vascular damage risk) and that deformations are physically meaningful.
neuroDeformNet: multimodal deformation network
The core contribution is MorphPINN, a multimodal network that fuses 2D X-ray image features with 3D geometric context to predict full 3D vessel deformation from a single fluoroscopy frame.
The architecture has three branches: 1. Vision Backbone — A ResNet-18 modified to accept single-channel (grayscale) X-ray images. Produces a 512-dimensional feature vector capturing vascular appearance and deformation cues. 2. 3D + Camera Encoder — A lightweight MLP that jointly encodes the 20 centerline control points in their rest configuration (60 values) and the 3×3 camera projection matrix (9 values) into a 128-dimensional geometric feature. 3. Fusion Predictor — The 512 image features and 128 geometric features are concatenated (640-dim) and passed through a 3-layer MLP with dropout, predicting a 20 × 3 displacement field — one 3D shift vector per control point.
physics-constrained loss
Training uses a composite loss that enforces both accuracy and physical plausibility:
\[\mathcal{L} = w_{\text{mse}} \cdot \mathcal{L}_{\text{MSE}} + w_{\text{smooth}} \cdot \mathcal{L}_{\text{smooth}} + w_{\text{proj}} \cdot \mathcal{L}_{\text{proj}}\]| Term | Weight | Purpose |
|---|---|---|
| \(\mathcal{L}_{\text{MSE}}\) | 1.0 | Penalizes deviation from ground-truth displacement |
| \(\mathcal{L}_{\text{smooth}}\) | 0.1 | Enforces deformation smoothness along the centerline |
| \(\mathcal{L}_{\text{proj}}\) | 1e-5 | Penalizes geometric inconsistency with the X-ray projection |
A hard strain cap of 1.15 is embedded in the loss, directly encoding the clinical safety constraint that prevents predictions from implying unsafe force thresholds on the vessel wall.
roadmap
- Physics-informed synthetic data generation (FFD + guidewire kinematics)
- Multi-view X-ray rendering pipeline (6 clinical projections via gVXR)
- Dataset physics auditing and validation
- NeuroDeformNet architecture and training pipeline
- Comprehensive literature review (under review, JIRS)
- Synthetic contrast injection to reduce chemical contrast agent dose
- Blood flow fluid dynamics integration for force threshold estimation
- Full intra-operative vessel motion prediction from guidewire state